
Your safety is our #1 priority. From time to time, certain prescription medications dispensed by Kinney Drugs may be recalled by the manufacturer(s). When this happens, we will display details on the recall(s), along with any instructions provided by the manufacturer(s) .
May 6th, 2026 -
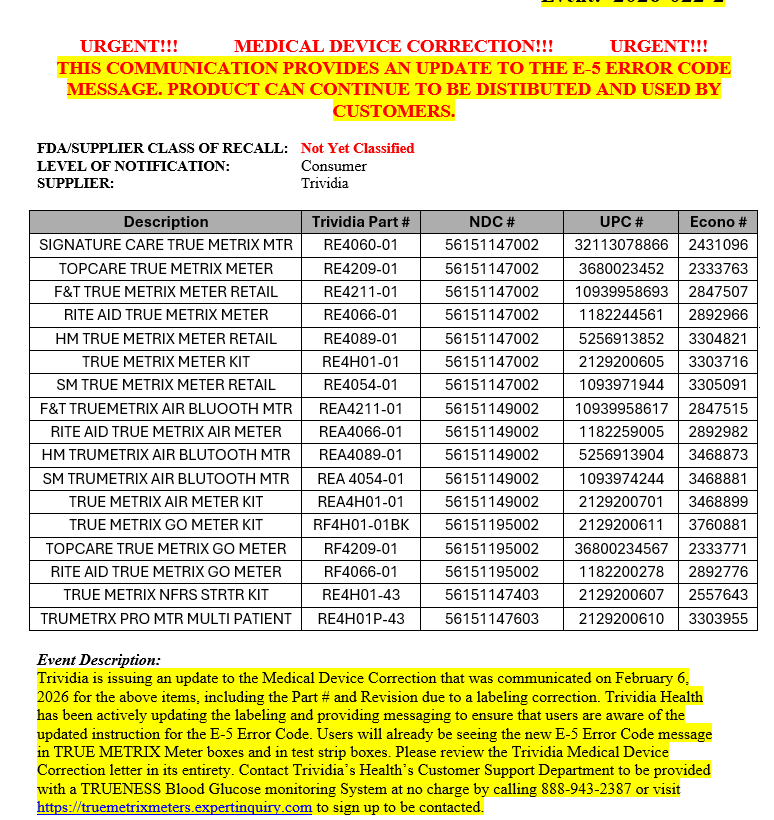
FDA/SUPPLIER CLASS OF RECALL: Not Yet Classified
LEVEL OF NOTIFICATION: Consumer
SUPPLIER: Trividia
|
Description |
Trividia Part # |
NDC # |
UPC # |
Econo # |
|
SIGNATURE CARE TRUE METRIX MTR |
RE4060-01 |
56151147002 |
32113078866 |
2431096 |
|
TOPCARE TRUE METRIX METER |
RE4209-01 |
56151147002 |
3680023452 |
2333763 |
|
F&T TRUE METRIX METER RETAIL |
RE4211-01 |
56151147002 |
10939958693 |
2847507 |
|
RITE AID TRUE METRIX METER |
RE4066-01 |
56151147002 |
1182244561 |
2892966 |
|
HM TRUE METRIX METER RETAIL |
RE4089-01 |
56151147002 |
5256913852 |
3304821 |
|
TRUE METRIX METER KIT |
RE4H01-01 |
56151147002 |
2129200605 |
3303716 |
|
SM TRUE METRIX METER RETAIL |
RE4054-01 |
56151147002 |
1093971944 |
3305091 |
|
F&T TRUEMETRIX AIR BLUOOTH MTR |
REA4211-01 |
56151149002 |
10939958617 |
2847515 |
|
RITE AID TRUE METRIX AIR METER |
REA4066-01 |
56151149002 |
1182259005 |
2892982 |
|
HM TRUMETRIX AIR BLUTOOTH MTR |
REA4089-01 |
56151149002 |
5256913904 |
3468873 |
|
SM TRUMETRIX AIR BLUTOOTH MTR |
REA 4054-01 |
56151149002 |
1093974244 |
3468881 |
|
TRUE METRIX AIR METER KIT |
REA4H01-01 |
56151149002 |
2129200701 |
3468899 |
|
TRUE METRIX GO METER KIT |
RF4H01-01BK |
56151195002 |
2129200611 |
3760881 |
|
TOPCARE TRUE METRIX GO METER |
RF4209-01 |
56151195002 |
36800234567 |
2333771 |
|
RITE AID TRUE METRIX GO METER |
RF4066-01 |
56151195002 |
1182200278 |
2892776 |
|
TRUE METRIX NFRS STRTR KIT |
RE4H01-43 |
56151147403 |
2129200607 |
2557643 |
|
TRUMETRX PRO MTR MULTI PATIENT |
RE4H01P-43 |
56151147603 |
2129200610 |
3303955 |
Event Description:
Trividia is issuing this Medical Device Correction for the above items, including the Part # and Revision due to a labeling correction that impacts the Owner’s Brooklets/System Instructions for Use included with the meter at Purchase and the online labeling and help guides at www.trividiahealth.com. The meter, test strips, and control solution are not impacted. The Products may continue to be used and sold. This correction does not require removal of the products from where they are used or sold. The correction involves the E-5 Error Code in the “Messages” section of the owner’s Booklets/System Instructions for use. The system displays an E-5 error code for a very high blood glucose event (>600mg/dl) or when there is a test strip error. Trividia is updating the E-5 error code actions to emphasize that users must seek medical attention immediately if they receive an E-5 error code and are experiencing symptoms of high glucose. This Medical device correction is to the Consumer level. Affected product started shipping August 1, 2014.
Consumer Information:
Patients should contact their doctor or health care provider for medical-related questions.
LEVEL OF NOTIFICATION: Consumer
SUPPLIER: Abbott - # 45290
|
Description |
Lot # |
UDI |
UPC # |
McKesson Item Number |
|
FREESTYLE LIBRE 3 SENSOR |
T60003054 |
00357599818005 |
35759981800 |
2638609 |
|
T60003085 |
||||
|
T60003088 |
||||
|
T60003089 |
||||
|
T60003092 |
||||
|
T60003099 |
||||
|
T60003113 |
||||
|
T60003136 |
||||
|
T60003159 |
||||
|
T60003160 |
||||
|
T60003271 |
||||
|
T60003284 |
||||
|
T60003374 |
||||
|
T60003375 |
||||
|
T60003391 |
||||
|
T60003426 |
||||
|
T60003434 |
||||
|
T60003507 |
||||
|
T60003534 |
||||
|
T60003546 |
||||
|
T60003547 |
||||
|
T60003564 |
||||
|
T60003628 |
||||
|
T60003646 |
||||
|
FREESTYLE LIBRE 3 PLUS SENSOR |
T60002984 |
00357599844004 |
35759984401 |
2967800 |
|
T60002994 |
||||
|
T60002995 |
||||
|
T60002996 |
||||
|
T60003007 |
||||
|
T60003009 |
||||
|
T60003010 |
||||
|
T60003138 |
||||
|
T60003243 |
||||
|
T60003255 |
||||
|
T60003348 |
||||
|
T60003352 |
||||
|
T60003366 |
||||
|
T60003369 |
||||
|
T60003377 |
||||
|
T60003378 |
||||
|
T60003379 |
||||
|
T60003384 |
||||
|
T60003398 |
||||
|
T60003400 |
||||
|
T60003404 |
||||
|
T60003446 |
||||
|
T60003449 |
||||
|
T60003454 |
||||
|
T60003455 |
||||
|
T60003460 |
||||
|
T60003470 |
||||
|
T60003471 |
||||
|
T60003473 |
||||
|
T60003478 |
||||
|
T60003479 |
||||
|
T60003480 |
||||
|
T60003481 |
||||
|
T60003488 |
||||
|
T60003490 |
||||
|
T60003491 |
||||
|
T60003492 |
||||
|
T60003494 |
||||
|
T60003496 |
||||
|
T60003497 |
||||
|
T60003498 |
||||
|
T60003518 |
||||
|
T60003542 |
||||
|
T60003543 |
||||
|
T60003544 |
||||
|
T60003550 |
||||
|
T60003552 |
||||
|
T60003560 |
||||
|
T60003562 |
||||
|
T60003563 |
||||
|
T60003567 |
||||
|
T60003568 |
||||
|
T60003570 |
||||
|
T60003573 |
||||
|
T60003575 |
||||
|
T60003576 |
||||
|
T60003585 |
||||
|
T60003589 |
||||
|
T60003592 |
||||
|
T60003596 |
||||
|
T60003601 |
||||
|
T60003611 |
||||
|
T60003617 |
||||
|
T60003618 |
||||
|
T60003619 |
||||
|
T60003631 |
||||
|
T60003639 |
||||
|
T60003641 |
||||
|
T60003683 |
Event Description:
Abbott is issuing an Urgent Medical Device Correction for the above items/lots due to incorrect low glucose readings. This notification is to the Consumer level. Affected product started shipping April 6, 2025.
Consumer Information:
To determine if your current sensor or any unused sensor(s) are potentially affected, please visit www.FreeStyleCheck.com and select “CONFIRM SENSOR SERIAL NUMBER.” You will need to locate your sensor serial number to determine if your sensor is potentially affected.
If you are currently wearing or have a FreeStyle Libre 3 / FreeStyle Libre 3 Plus sensor that has been confirmed as potentially affected on www.FreeStyleCheck.com or by a customer service representative, immediately discontinue use and dispose of the affected sensor(s).
You can request a replacement for any potentially affected sensor(s) on www.FreeStyleCheck.com. Select “CONFIRM SENSOR SERIAL NUMBER” and enter a valid serial number. If your sensor is potentially impacted, you will be instructed to enter your contact information so a replacement product can be sent to you at no cost.
September 19th, 2025 -
SUPPLIER: Dr. Reddy’s - 19991
|
Description |
Lot # |
Exp Date |
NDC # |
UPC # |
Econo # |
|
ROPINIR XR TAB 2MG DR/R 90@ |
C2315044 |
9/30/2025 |
55111065990 |
35511165990 |
1176908 |
|
ROPINIR XR TAB 2MG DR/R 30@ |
C2315045 |
9/30/2026 |
55111065930 |
35511165930 |
1176098 |
Event Description:
Dr. Reddy’s is voluntarily withdrawing the above items/lots due to the Out of Trend result observed during the 18-month stability review of related substance data. This withdrawal is to the Consumer level. Affected product started shipping December 15, 2023.
Consumer Information:
Patients should contact their doctor or health care provider for medical-related questions.
June 4th, 2025 -
LEVEL OF NOTIFICATION: Consumer
SUPPLIER: Amneal - # 12853
|
Description |
Lot # |
Exp Date |
NDC # |
UPC # |
Econo # |
|
SULFAM+TRI TB 400/80MG AMN100@ |
AM241019 |
6/30/2027 |
65162027110 |
36516227110 |
3990959 |
|
AM241020 |
6/30/2027 |
||||
|
SULFAM+TRI TB400/80MG AMN 500@ |
AM241019A |
6/30/2027 |
65162027150 |
36516227150 |
3990967 |
Event Description:
Amneal is voluntarily recalling the above items/lots due to tablets that may exhibit black spots due to microbial contamination. This recall is to the Consumer level. Affected product started shipping December 4, 2024.
Consumer Information:
Patients should contact their doctor or health care provider for medical-related questions.
May 28th, 2025 -
LEVEL OF NOTIFICATION: Consumer
SUPPLIER: Teva- # 26108
|
Description |
Lot # |
Exp Date |
NDC # |
UPC # |
Econo # |
|
METOCLOPR TAB 10MG TEV 100@ |
5420094 |
9/30/2027 |
93220301 |
30093220301 |
1813708 |
Event Description:
Teva is voluntarily recalling the above item/lot due to a single Torsemide Tablet (20mg) was discovered in each of three sealed bottles of Metoclopramide tablets. This recall is to the Consumer level. Affected product started shipping December 16, 2024.
Consumer Information:
Patients should contact their doctor or health care provider for medical-related questions.
May 16th, 2025 -
URGENT: Medical Device Correction
Dexcom G6 Receive - MT27408-1, Dexcom G7 Receive - MT26403-0
| Part Number | SKUs | Part Description |
| MT27408-1 | STK-FE-001, STK-FM-001, STK-FR-001 | Dexcom G6 Receiver |
| MT26403-0 | STK-AT-011, STK-AT-012, STK-AT-013 | Dexcom G7 Receiver |
Description of the Problem:
Users of the Dexcom G6 or G7 Receiver have reported an issue in which the receiver may not provide audio output to alerts/alarms as expected.
April 30th, 2025 -
FDA/SUPPLIER CLASS OF RECALL: Not Yet Classified
LEVEL OF NOTIFICATION: Consumer
SUPPLIER: AVKARE - # 04522, 04582
|
Description |
Lot # |
Exp Date |
NDC # |
Econo # |
|
ARTIFICIAL TEAR OPH/O AVK 15ML |
126 |
10/26/2025 |
50268004315 |
2821411 |
|
127 |
10/30/2025 |
|||
|
128 |
11/2/2025 |
|||
|
129 |
11/6/2025 |
|||
|
162 |
5/9/2026 |
|||
|
163 |
5/14/2026 |
|||
|
164 |
5/20/2026 |
|||
|
165 |
5/23/2026 |
|||
|
166 |
5/29/2026 |
|||
|
167 |
6/3/2026 |
|||
|
168 |
6/6/2026 |
|||
|
169 |
6/10/2026 |
|||
|
170 |
6/13/2026 |
|||
|
193 |
10/7/2026 |
|||
|
194 |
10/10/2026 |
|||
|
195 |
10/14/2026 |
|||
|
196 |
10/17/2026 |
|||
|
197 |
10/21/2026 |
|||
|
198 |
10/24/2026 |
|||
|
199 |
10/30/2026 |
|||
|
CARBOXY SOD OPH GEL 1% AVK15ML |
114 |
9/4/2025 |
50268006615 |
2635233 |
|
115 |
9/6/2025 |
|||
|
116 |
9/10/2025 |
|||
|
207 |
12/5/2026 |
|||
|
CARBOXYM SOD OP/S 0.5%AVK15ML@ |
103 |
4/26/2025 |
50268006815 |
2304061 |
|
104 |
5/3/2025 |
|||
|
108 |
6/29/2025 |
|||
|
109 |
7/18/2025 |
|||
|
110 |
8/17/2025 |
|||
|
111 |
8/22/2025 |
|||
|
112 |
8/27/2025 |
|||
|
113 |
9/18/2025 |
|||
|
125 |
10/23/2025 |
|||
|
130 |
11/9/2025 |
|||
|
131 |
11/14/2025 |
|||
|
132 |
11/27/2025 |
|||
|
133 |
11/30/2025 |
|||
|
134 |
12/5/2025 |
|||
|
135 |
12/11/2025 |
|||
|
136 |
12/14/2025 |
|||
|
137 |
1/2/2026 |
|||
|
139 |
1/15/2026 |
|||
|
140 |
1/19/2026 |
|||
|
141 |
1/25/2026 |
|||
|
151 |
3/18/2026 |
|||
|
152 |
3/21/2026 |
|||
|
153 |
3/25/2026 |
|||
|
154 |
3/28/2026 |
|||
|
155 |
4/8/2026 |
|||
|
156 |
4/8/2026 |
|||
|
157 |
4/11/2026 |
|||
|
160 |
4/26/2026 |
|||
|
180 |
8/8/2026 |
|||
|
181 |
8/12/2026 |
|||
|
182 |
8/18/2026 |
|||
|
183 |
8/21/2026 |
|||
|
184 |
8/26/2026 |
|||
|
185 |
9/4/2026 |
|||
|
186 |
9/9/2026 |
|||
|
187 |
9/16/2026 |
|||
|
189 |
9/21/2026 |
|||
|
190 |
9/25/2026 |
|||
|
191 |
9/28/2026 |
|||
|
192 |
10/2/2026 |
|||
|
208 |
12/11/2026 |
|||
|
209 |
12/16/2026 |
|||
|
212 |
1/15/2027 |
|||
|
213 |
1/21/2027 |
|||
|
214 |
1/24/2027 |
|||
|
215 |
2/2/2027 |
|||
|
216 |
2/4/2027 |
|||
|
224 |
3/27/2027 |
|||
|
POLYET GLY OPT SOL400 AVK 15ML |
117 |
9/20/2025 |
50268012615 |
2823375 |
|
118 |
9/25/2025 |
|||
|
119 |
9/27/2025 |
|||
|
121 |
10/5/2025 |
|||
|
161 |
5/1/2026 |
|||
|
171 |
6/18/2026 |
|||
|
172 |
6/24/2026 |
|||
|
174 |
7/1/2026 |
|||
|
175 |
7/8/2026 |
|||
|
200 |
11/5/2026 |
|||
|
201 |
11/10/2026 |
|||
|
202 |
11/13/2026 |
|||
|
203 |
11/18/2026 |
|||
|
204 |
11/21/2026 |
|||
|
205 |
11/25/2026 |
|||
|
206 |
12/2/2026 |
|||
|
219 |
2/24/2027 |
|||
|
221 |
3/2/2027 |
|||
|
222 |
3/5/2027 |
|||
|
POLYVINYL OPH/SOL 1.4% AVK15ML |
120 |
10/2/2025 |
50268067815 |
2821429 |
|
122 |
10/9/2025 |
|||
|
123 |
10/12/2025 |
|||
|
124 |
10/16/2025 |
|||
|
138 |
10/8/2026 |
|||
|
142 |
12/9/2026 |
|||
|
143 |
2/1/2026 |
|||
|
144 |
2/7/2026 |
|||
|
145 |
2/12/2026 |
|||
|
146 |
2/15/2026 |
|||
|
147 |
2/21/2026 |
|||
|
148 |
2/27/2026 |
|||
|
149 |
3/4/2026 |
|||
|
150 |
3/11/2026 |
|||
|
158 |
4/15/2026 |
|||
|
159 |
4/22/2026 |
|||
|
176 |
7/24/2026 |
|||
|
177 |
7/28/2026 |
|||
|
178 |
7/31/2026 |
|||
|
179 |
8/5/2026 |
Event Description:
AVKARE is voluntarily recalling the above items/lots due to manufacturing cGMP deviations identified during an audit by the FDA. This recall is to the Consumer level. Affected product started shipping May 26, 2023.
Consumer Information:
Patients should contact their doctor or health care provider for medical-related questions.
March 14th, 2025 -
LEVEL OF NOTIFICATION: Consumer
SUPPLIER: Exela - # 15287
|
Description |
Lot # |
Exp Date |
NDC # |
UPC # |
Econo # |
|
SOD BIC SDV 50MEQ/50ML EXE 25@ |
10006417 |
10/1/2026 |
51754500104 |
35175450014 |
2630101 |
|
10006418 |
10/1/2026 |
Event Description:
Exela is voluntarily recalling the above item/lots due to the complaints that some vials had an insufficient crimp on the cap and stopper which may expose the rug product to the environment and could potentially impact the sterility and purity of the product. This recall is to the Consumer level. Affected product started shipping February 5, 2025.
Consumer Information:
Patients should contact their doctor or health care provider for medical-related questions.